Product simulation for pregnancy diagnostics
Sector: Medical technologySpecialist field: Structural mechanicsPregnolia supports the risk assessment of premature births with its medical technology products. Structural-mechanical simulation identifies critical parameters and paves the way for large-scale production.
Summary
Task
Pregnolia develops medical measuring devices to improve pregnancy care. When switching to large-scale production, the manufacturing process for the probe cap is to be changed from milling to injection moulding without the new manufacturing tolerances affecting the measurement accuracy.
Solution
Transient structural mechanics simulations determine the accuracy of the pressure measurement with respect to the geometry tolerances from the manufacturing process. With this sensitivity study, the measurement accuracy was successfully confirmed taking into account the new wider manufacturing tolerances.
Customer benefits
The analyses contributed to a significant reduction in prototypes and tests for elaborate injection moulds, with clear time and cost savings. The time span for the conversion to efficient large-scale production while complying with all critical measurement criteria despite changed manufacturing tolerances is thus significantly reduced.
Project Details
Task
A WHO survey from 2016 shows that more than 15 million babies are born prematurely every year. The occurrence of preterm birth is complex and can have many causes. Research has shown that a key indicator of the risk of preterm birth is the firmness of the cervix.
Pregnolia, a spin-off of ETH Zurich, develops medical measuring devices to improve pregnancy care. When switching to large-scale production, the manufacturing for the probe cap is to be changed from plastic milling to injection moulding without the new manufacturing tolerances having an effect on the measurement accuracy.
In order to evaluate the influence of these tolerance changes on the measurement accuracy of the probe, a sensitivity study should be carried out. Real cervical measurement data are available to validate the simulation results.
Customer Benefit
With the help of the sensitivity analysis, the critical manufacturing tolerances could be identified. Validation measurements with prototypes of the probe cap also confirmed the validity of the selected parameters and the accuracy of the simulation results. These findings can now be used for a targeted improvement of the manufacturing tolerances, which in turn leads to an increased accuracy of the Pregnolia diagnostic device.
These analyses contributed to a significant reduction in the number of prototypes and tests required, which means clear time and cost savings. The time required for a safe transition to efficient and mature large-scale production was significantly reduced. The Pregnolia measuring device is a valuable support in preterm birth diagnostics. With large-scale production, high demand can be met more quickly.
At the same time, these tests reinforce confidence in the measuring principle and in the accuracy of the results. Results that help to protect life.
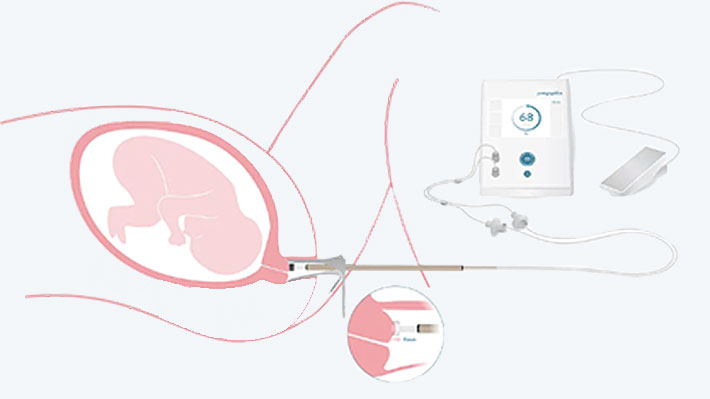
Solution
An increasingly negative pressure is generated in the measuring device, which sucks tissue into the probe cap. When a defined suction height is reached, the necessary pressure is measured. It is considered an indicator of tissue stiffness.
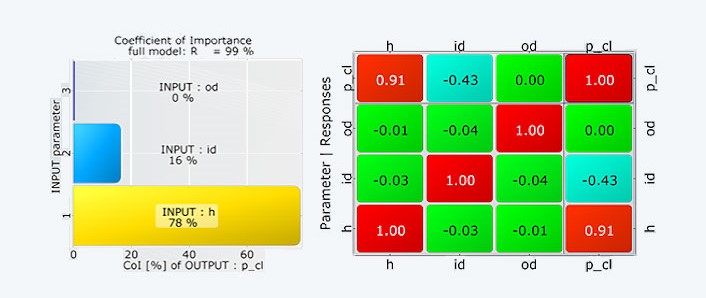
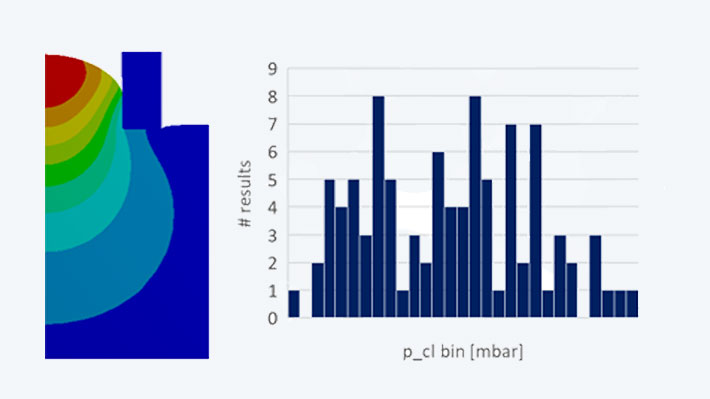
The simulation model is based on an axially symmetrical model of the probe cap. The parameters of the hyperelastic material were calibrated with the real cervical tissue parameters. In a transient structural simulation, an identical pressure-time curve is defined as the load. The decisive investigation parameter is the sensitivity of the measured pressure when reaching the suction height with regard to the fabric-dependent geometric dimensions.
The evaluations showed that within the new manufacturing tolerances the critical value, which determines the accuracy of the measuring principle, only varies by +/- 3.7%. The required reliability of the measurement is thus safely achieved.
Images: © pregnolia




